CHE 318 Lecture 28
Humidification Process II
2026-03-18
Learning outcomes
After this lecture, you will be able to:
- Recall humidification and cooling paths on the psychrometric chart.
- Describe the origin of wet-bulb temperature and latent-heat effects during evaporation.
- Analyze adiabatic saturation using coupled heat and mass balances.
- Apply the psychrometric chart to obtain humidity-related properties.
Cheatsheet for humidification process
Charts distributed in class.
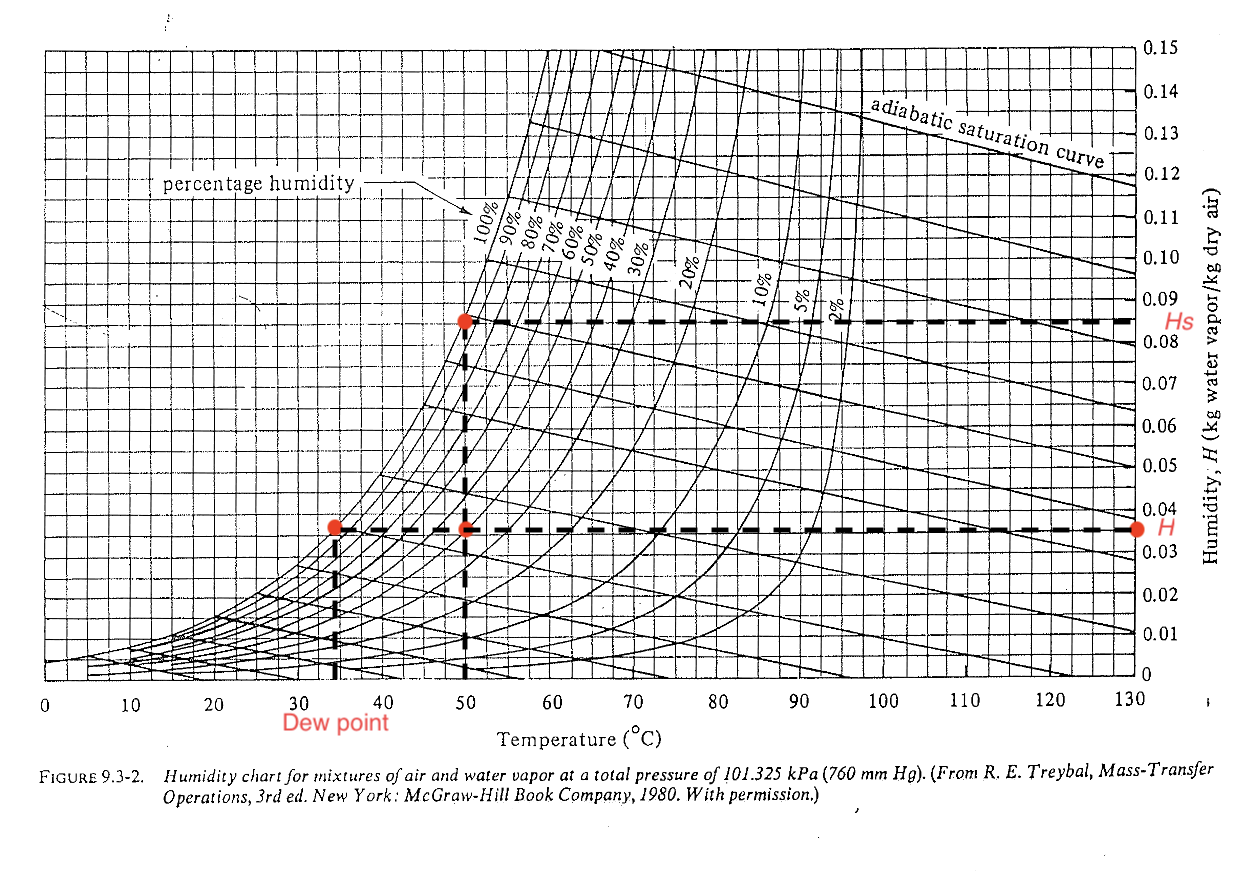
Reading the humidity / psychrometric chart (level 1)
Find where \(H\), \(H_p\), dew point \(T_{\text{dew}}\) are?
Example 2: humidifier design
A living room of 60 m\(^2\) at 22 \(^\circ\)C has a relative humidity of 20% due to continuous heating. You and your roommate wish to purchase a humidifier that can humidify the living room up to 45% relative humidity. Assume the floor-ceiling distance is 3 m, calculate the weight of water needed to humidify the whole room. Can you use the humidity chart to estimate?
Example 2: steps
- Read the saturation vapour pressure from \(H_s\) (\(H_s = 0.017\))
from the vapour data it gets \(p_{\text{vap}} = 2.64\) kPa, pretty close!
- Estimate weight of water \(\Delta m_A\)
Does it make sense? The requirement for water tank will be a lower bound.
Mythbusting time (1)
My weather app shows that outside is \(-12\ ^\circ\)C and R.H. is 74%, where the dew point is \(-16\ ^\circ\)C. What does all that mean?
Tip
A useful note is that the lower dew point is compared with current temperature, the drier air appears to be
Mythbusting time (2)
In the previous scenario \(-12\ ^\circ\)C and R.H. is 74%, when I open the door, my humidity in home immediately drops! Why?
- The calculated \(H_R\) at room temperature is only 6.7% if fully filled with outside air.
Tip
\(H_R\) (or R.H.) only measures up at current temperature. When moving water content at different \(T\), \(H_R\) will change.
- cold air 👉 appears to be dry at high T
- warm air 👉 appears to be humid at low T
Mythbusting time (3)
Why does “humid hot” environment feels much hotter than “dry hot” environment, even if the apparent \(T\) is the same?
- Calculate the enthalpy of air-water mixture, \(H_y\) significantly increases when relative humidity is high!
- Human body relies on cooling to survive. At high \(T\) high \(H_R\), both driving forces for evaporation are reduced:
- \(T_{\text{body}} - T_{\text{env}}\) becomes small 👉 bad dissipation
- \(p_{A, \text{body}} - p_{A, \text{env}}\) becomes small 👉 evaporation suppressed
Tip
The sensible specific heat for air-water mixture does not change significantly, even if water vapour’s heat capacity is higher. (absolute \(H\) is always 0.1 when \(T<50\ ^\circ\)C)
Measuring humidity: the two-bulb hygrometer
Before electronic sensors, humidity was measured using a two-thermometer setup
- One thermometer measures dry-bulb temperature \(T_d\)
- The second thermometer is wrapped with a wet wick and measures wet-bulb temperature \(T_w\) (\(T_w < T_d\))
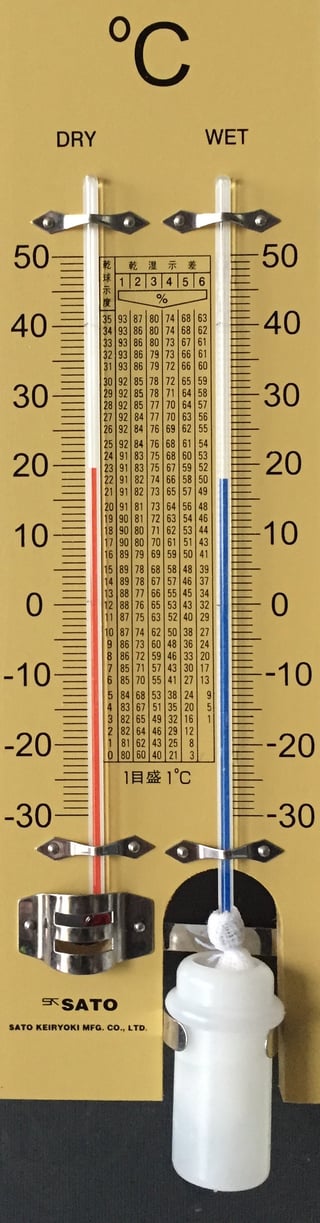
What does the wet bulb tell us?
The wet bulb is a steady state evaporative cooling experiment, which can be generalized for interfacial evaporation problem:
- In and outlet temperatures are the same, while outlet humidity is higher
- Evaporation consumes latent heat
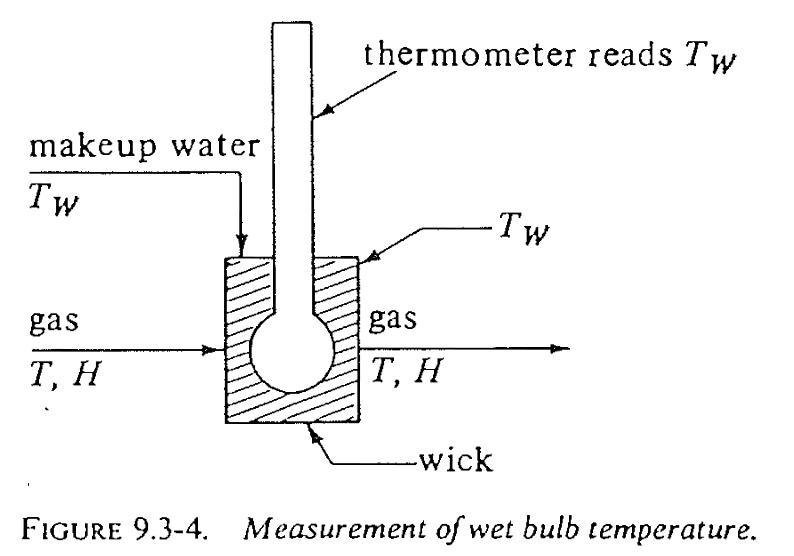
Concept of wet-bulb temperature
- The wet-bulb temperature \(T_w\) is the temperature reached by a wet surface exposed to air
\[ T_w < T_d \]
- \(T_d\) = dry-bulb temperature
- \(T_w\) = wet-bulb temperature
- The difference between \(T_w\) and \(T_d\) indicates the inlet air humidity
Moist air enthalpy
From Lecture 27, the specific enthalpy of moist air can be written as
\[\begin{align} H_{y} = (1.005 + 1.88H)(T - T_0) + 2501.4H \end{align}\]- Increase the absolute humidity \(H\) must cause \(H_y\) to increase 👉 gas phase takes heat from liquid.
- Solving the heat + mass balance will give solution to \(T_w\).
Heat balance around the wet bulb
Consider the control volume around the wet bulb. Heat transferred from the air is used to evaporate water, linked by the heat-mass balance (need a bit prerequisite in heat transfer):
\[\begin{align} q = M_A \lambda_w A N_A \end{align}\]- \(m_A\): molecular weight of water
- \(\lambda_w\): latent heat of vaporization at \(T_w\) (44045 kJ/kg mol at 1 atm)
- \(A\): area of the wetted surface
- \(N_A\): molar flux of evaporating water
R.H.S.: mass transfer at the interface
The evaporation rate can be written as
\[\begin{align} N_A = k_y (y_w - y) \end{align}\]- \(k_y\): gas-phase mass transfer coefficient (since \(y_{BM} \approx 1\), \(k_y \approx k_y'\))
- \(y_w\): vapor mole fraction at the interface (saturated at \(T_w\))
- \(y\): vapor mole fraction in bulk air
Converting humidity to mole fraction
We can further change \(y\) to the humidity \(H\) for dilute system. Since humidity is define as weight ratio:
\[\begin{align} H = \frac{\text{kg }H_2O}{\text{kg dry air}} \end{align}\]The mole fraction of vapor is
\[\begin{align} y = \frac{H/M_A}{1/M_B + H/M_A} \end{align}\]Since humidity is typically small
\[\begin{align} y \approx \frac{M_B}{M_A}H \end{align}\]L.H.S. heat transfer to the wet surface
The heat flux \(q\) in L.H.S. from the air to the wet surface is
\[\begin{align} q = h (T - T_w) A \end{align}\]where \(h\) is heat transfer coefficient in the Fourier’s law \(q = -h \Delta T\)
Wet-bulb setup: final results
Combining heat transfer (\(q\)) and mass transfer (\(N_A\)) relations gives
\[\begin{align} \frac{H - H_w}{T - T_w} = - \frac{h}{M_B k_y \lambda_w} \end{align}\]Note \(\frac{H - H_w}{T - T_w}\) means the slope of a line on the psychrometric chart. The slope is almost identical to adiabatic line!
Psychrometric chart: adiabatic line (level 2)
The \((T_d, H_{\text{in}})\) and \((T_w, H_{\text{out}})\) points are along the adiabatic line (no external heat exchange). For water-air system, the adiabatic line and cooling line are very close and often not distinguished.
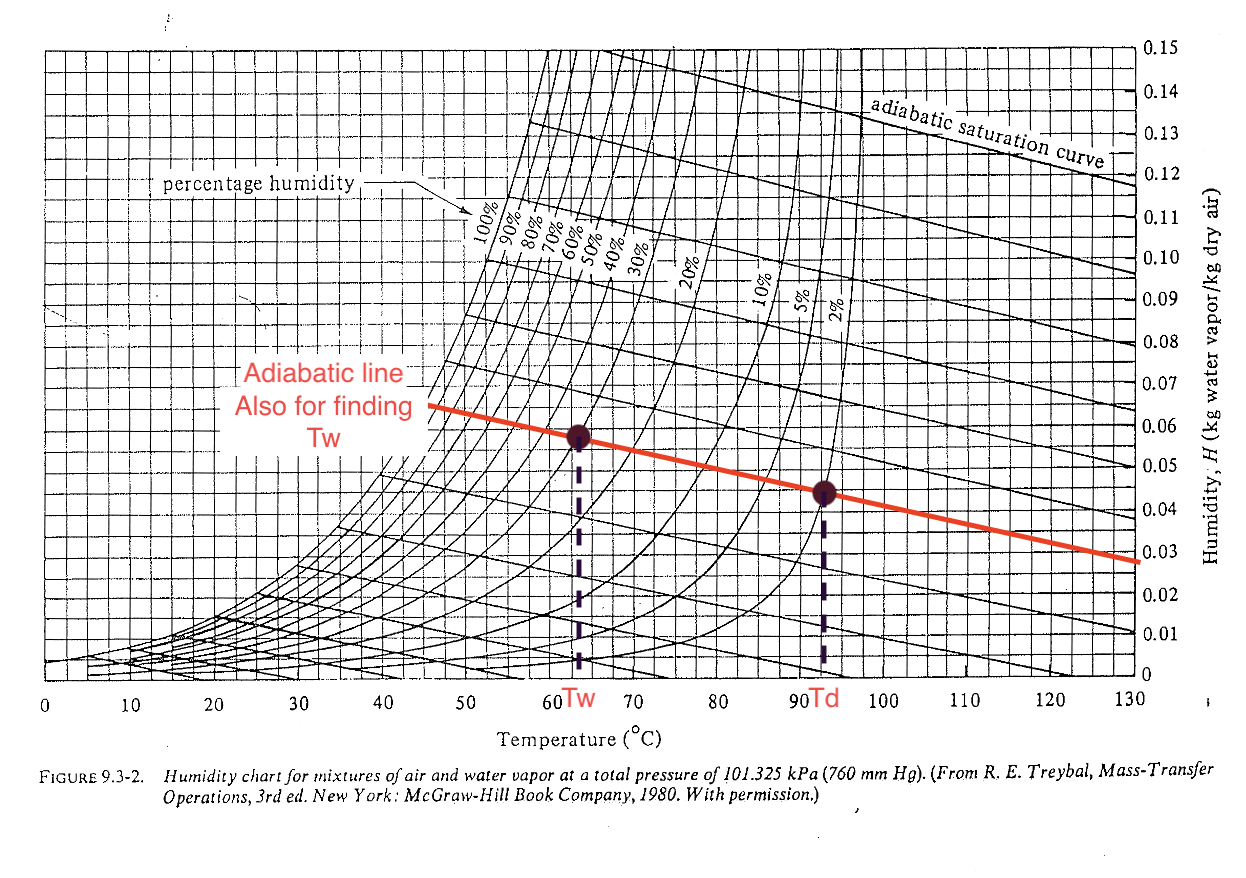
Deeper look into the cooling process (1)
What does the adiabatic line tell us? It is basically a process that each point has the same humid enthalpy, and no change of heat to external system:
\[ H = c_s (T - T_0) + H \lambda_0 = \text{[Const]} \]
- Increase humidity ➡ decrease \(T\)
- Lowest temperature can reach in the system at certain \(H_{\text{out}}\) is \(T_w\)
- Lowest temperature can reach when air is saturated is \(T_s\)
Deeper look into the cooling process (2)
For water-air, one handy property is that
\[ \frac{h}{M_B k_y} \approx 1.005 \approx c_s \qquad \text{[kJ / kg air]} \]
such relation allows us to use the humidity chart’s adiabatic saturation curve.
Warning
Such simplification may not be applicable for other liquid, such as benzene!
What to learn next
The wet-bulb experiment connects heat transfer and mass transfer, and the evaporation rate depends on the vapor pressure driving force.
Next topics we will study:
- Gas-phase mass transfer during evaporation
- Interfacial equilibrium between water and air
- Driving force expressed as \((y_w - y)\) or \((p_{sat} - p_A)\)
Summary
- Reading humidity chart for cooling process
- Wet-bulb temperature and its origin
- Calculation of humidity values from the chart + equation