Interface concentrations: x_Ai = 0.246589, y_Ai = 0.183180
N_A from gas film: 2.883412e-04 (same units as k_y)
N_A from liquid film: 2.883412e-04 (same units as k_x)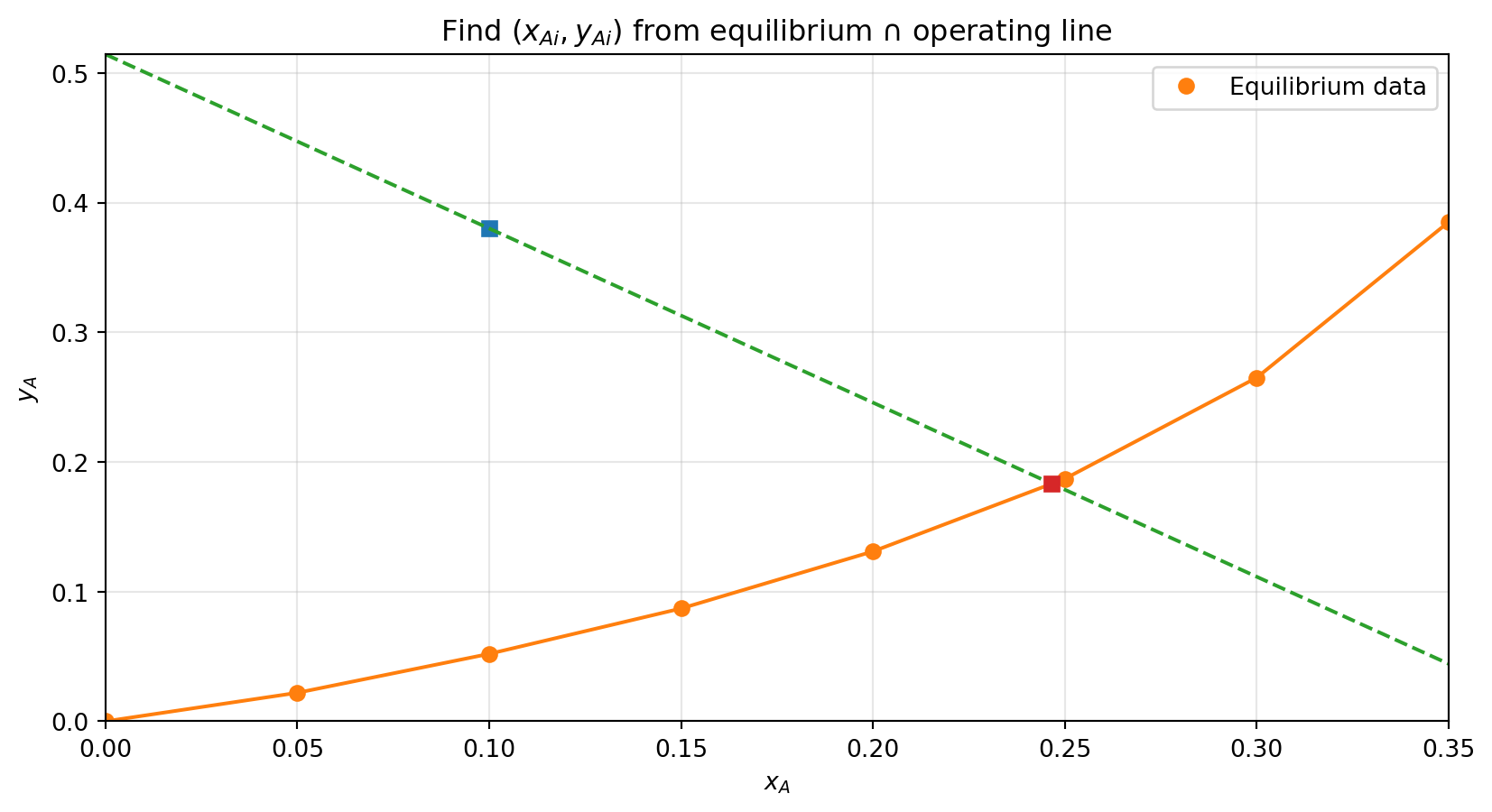
Interface Mass Transfer
2026-03-04
After this lecture, you will be able to:
The most complex case is probably a packed-bed column.
We we have focused on?
What we may miss?
Absorption tower
Extraction apparatus (liquid-gas)
Extraction apparatus (liquid-liquid)
Overall mass balance between liquid and gas
\[\begin{align} \text{In}_{\text{liq}} + \text{In}_{\text{gas}} = \text{Out}_{\text{liq}} + \text{Out}_{\text{gas}} \end{align}\]In each phase, we can use our knowledge from packed bed lecture, e.g.
\[\begin{align} \text{In}_{\text{gas}} - \text{Out}_{\text{gas}} + \text{Gen}_{\text{gas}} &= 0 \\ Q (c_1 - c_2) + A_{\text{eff}} \hat{N}_{\text{eff}} &= 0 \end{align}\]In Lecture 14 we discussed the interfacial concentration and mass balance. We need to know 1) The equilibrium constant \(K\) at the interface 2) The ratio between \(k_c'\) in two phases
Most commonly in industry we can use the equilibrium plot between A’s molar fractions in gas \(y_A\) (or \(p_A\)) and liquid \(x_A\), respectively.
Simpliest situation is Henry’s law
\[ p_A = H x_A \]
Meaning of points on the equilibrium curve – interfacial concentraion
A solute is being absorbed from a gas mixture of A and B in a wetted-wall tower, with the liquid flowing downwards. At a certain point in the tower, the bulk gas concentration of A is \(y_{AG}=0.380\) and the bulk liquid fraction is \(x_{AL}=0.100\). The film transfer coefficients for A in gas and liquid phases are: \(k_y'=1.465\times 10^{-3}\) kg mol/m\(^2\)/s and \(k_x'=1.967\times 10^{-3}\) kg mol/m\(^2\)/s. You can assume the \(k_x' \approx k_x\) and \(k_y' \approx k_y\). The following pairs of equilibrium \((x_Ai, y_Ai)\) data were measured:
\((0.0, 0.0), (0.05, 0.022), (0.10, 0.052) (0.15, 0.087)\)
\((0.20, 0.131), (0.25, 0.187), (0.30, 0.265), (0.35, 0.385)\)
Interface concentrations: x_Ai = 0.246589, y_Ai = 0.183180
N_A from gas film: 2.883412e-04 (same units as k_y)
N_A from liquid film: 2.883412e-04 (same units as k_x)