MATE 664 Lecture 15
Heterogeneous Nucleation
- Slides 👉 Open presentation🗒️
- PDF version of course note 👉 Open in pdf
- Handwritten notes 👉 Open in pdf
Learning outcomes
After this lecture, you will be able to:
- Compare heterogeneous nucleation with homogeneous nucleation
- Analyze the driving-force terms in both cases
- Derive the ratio between heterogeneous and homogeneous nucleation barriers and rates
- Recall methods for determining the equilibrium nucleation shape
Recall: key results from homogeneous nucleation
Homogeneous (spherical) nucleus gives the following results:
- Critical nucleus size
\[ n_c = -\frac{8}{27} \left[\frac{\eta \gamma_{\alpha \beta}}{\mu_\beta - \mu_\alpha} \right]^3 \]
- Nucleation free energy barrier
\[ \Delta G_c = \frac{4}{27} \frac{(\eta \gamma)^3}{(\mu_\beta - \mu_\alpha)^2} \]
- Nucleation rate
Recall: homogeneous nucleation implications
- \(\Delta G_c \propto \gamma^3\): very sensitive to the interfacial free energy
- Zeldovich factor \(Z\) is around 0.1
- Particles can shrink when they are not reaching \(n_c\)!
- Rule of thumb: \(\Delta G_c \leq 76 k_B T\), otherwise no detectable nucleation
- At \(T=298\) K, \(\Delta G_c \leq 1.95\) eV
What’s the missing picture?
Can we really treat the nuclei as spheres?
Practical considerations: heterogeneous nucleation
- General idea: can we have smaller \(\Delta G_c\) if other competing energies exist in the system?
- Analog: nucleation of crystals on a beaker wall
- Need to consider the free energy change before and after wall surface is covered by the nucleus
Triple interface balance: the Young’s equation
Analogous to the classical wetting theory, the “contact angle” on a droplet can be described by the Young’s equation
\[ \gamma_{\mathrm{S}} = \gamma_{\mathrm{L}} \cos \theta + \gamma_{\mathrm{SL}} \]
Heterogeneous nucleation on a wall: volume vs surface
The geometry of the droplet gives:
- Volume of nucleus: \(V_{\mathrm{n}} = \frac{\pi R^{3}}{3} (2 - 3 \cos \theta + \cos^{3} \theta)\)
- Interfacial area of nucleus with solution: \(A_{\mathrm{s}} = 2 \pi R^{2} (1 - \cos \theta)\)
- Interfacial area of nucleus with wall: \(A_{\mathrm{c}} = \pi R^{2} \sin^{2} \theta\)
Final solution to heterogeneous nucleation barrier:
\[ \Delta G_c^{\text{het}} = V_{\mathrm{n}} \Delta G_{\mathrm{V}} + \gamma_{\mathrm{sn}} A_{\mathrm{s}} + (\gamma_{\mathrm{nw}} - \gamma_{\mathrm{sw}}) A_{\mathrm{c}} \]
Heterogeneous nucleation on a wall: results
We can compare the hetero- and homogeneous barriers:
\[ \frac{\Delta G_{\mathrm{het}}}{\Delta G_{\mathrm{homo}}} = \frac{2 - 3 \cos \theta + \cos^{3} \theta}{4} = f \]
Heterogeneous in binary alloys: geometry
- At triple-interface, we have the balance
\[ \gamma_{\alpha\alpha} = 2 \gamma_{\alpha \beta} cos \psi \]
- Grain boundary \(\gamma_{\alpha\alpha} \neq 0\)
- What does \(\gamma_{\alpha\alpha} = 0\) mean? Homogeneous nucleation!
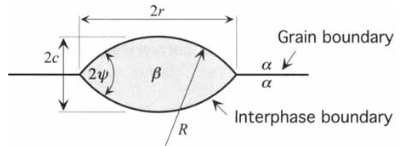
Heterogeneous in binary alloys: \(\Delta G_c^{\text{het}}\) results

- Volume \(V = \frac{2 \pi R^3}{3} (2 - 3 \cos \psi + \cos^3 \psi)\)
- Area of cap \(A_c = 4 \pi R^2 (1 - \cos \psi)\)
- Area below the cap: \(A_b = \pi r^2 = \pi R^2 (1 - \cos^2 \psi)\)
Overall heterogeneous nucleation barrier:
\[ \Delta G_c^{B} = (\frac{2 \pi R^3}{3} \Delta G_m + 2\pi R^2 \gamma_{\alpha\beta})(2 - 3 \cos \psi + \cos^3 \psi) \]
Ratio between hetero- and homogeneous nucleation energy barriers
Compare the two barriers, they are quite similar
\[\begin{align} \Delta G_c^{H} &= (\frac{4 \pi R^3}{3} \Delta G_m + 4\pi R^2 \gamma_{\alpha\beta}) \\ \Delta G_c^{B} &= (\frac{2 \pi R^3}{3} \Delta G_m + 2\pi R^2 \gamma_{\alpha\beta})(2 - 3 \cos \psi + \cos^3 \psi) \end{align}\]Ratio:
\[\begin{align} \frac{\Delta G_c^{B}}{\Delta G_c^{H}} = \frac{1}{2}(2 - 3 \cos \psi + \cos^3 \psi) \end{align}\]This is similar to our case of heterogeneous nucleation on a wall, but with different coefficient (why?)!
Heterogeneous nucleation barrier
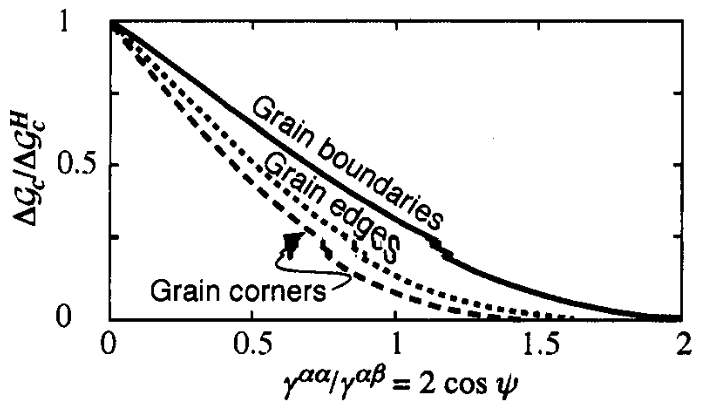
Heterogeneous nucleation in alloys: other nucleus dimensionalities
- From previous figure we see that \(\Delta G_c\) on defect becomes smaller for lower-dimensionality defects
- But do low dimensional defects always win? Not essentially.
- Number of sites available also decreases.
Assume the average grain size is \(L\), with grain boundary thickness \(\delta\), available sites follows
\[ n^{\text{defect}} \propto n_t (\frac{\delta}{L})^{3 - d} \]
Competition between hetero- and homogeneous nucleation rates
When considering the rates, two factors matter in the overall \(J\) equation:
- Free energy barrier \(\exp( - \Delta G_c/k_B T)\) (hetero > homo)
- Total available sites \(n_t\) (hetero < homo)
\[ \ln\!(\frac{J^B}{J_H}) = \ln\!(\frac{\delta}{L}) + \frac{\Delta G_c^H - \Delta G_c^B}{k_B T} \]
Overall nucleation regimes
- \(R_B = k_B T\ln(\frac{L}{\delta})\)
- Homogeneous nucleation favours when \(R_B > \Delta G_c^H - \Delta G_c^B\)
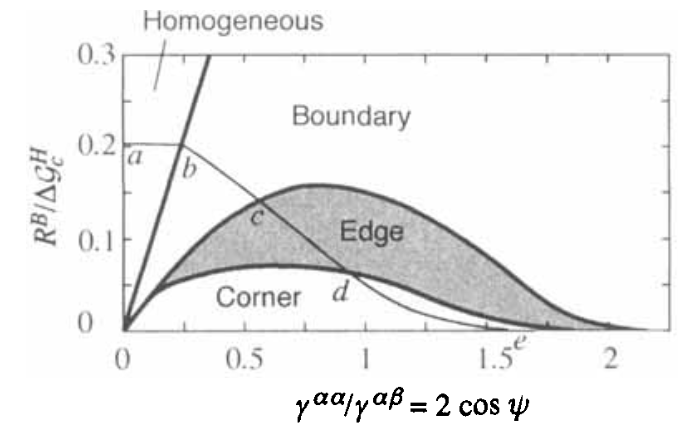
What else may be missing?
- The nucleation geometry may be very different from spherical or curved!
- Different facets have distinct surface free energy
- Overall goal: when the volume \(V\) is fixed, can we know the equilibrium shape of a crystal, so that surface energy is minimized?
- Wulff construction: optimizing the geometry of equilibrium shape
- Higher energy facet would have longer distance to the center!
Wulff construction for equilibrium crystal shape
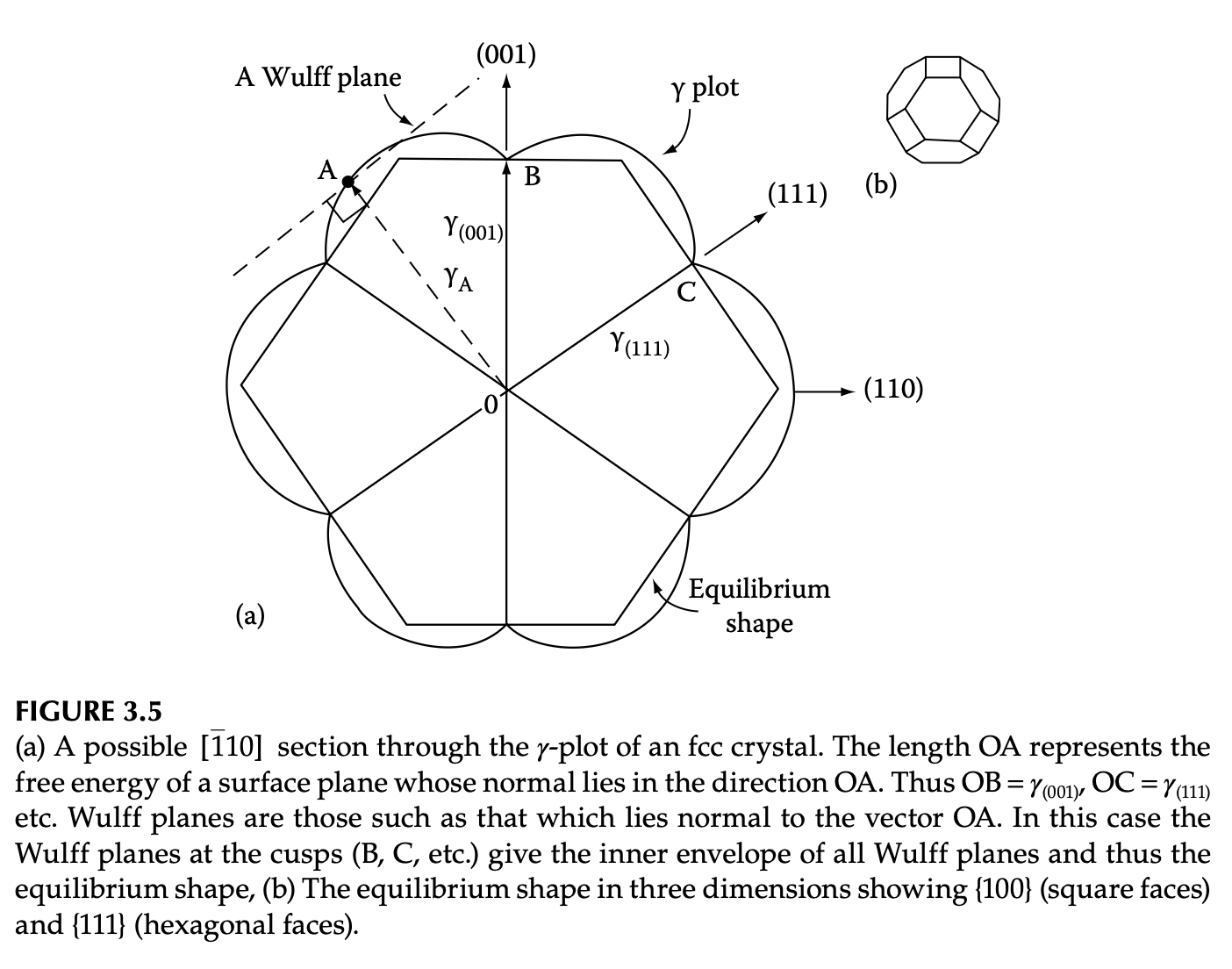
Theoretical Wulff construction for elements
Tran et al. Scientific Data, 2016, 3:160080
Nucleation: example demo
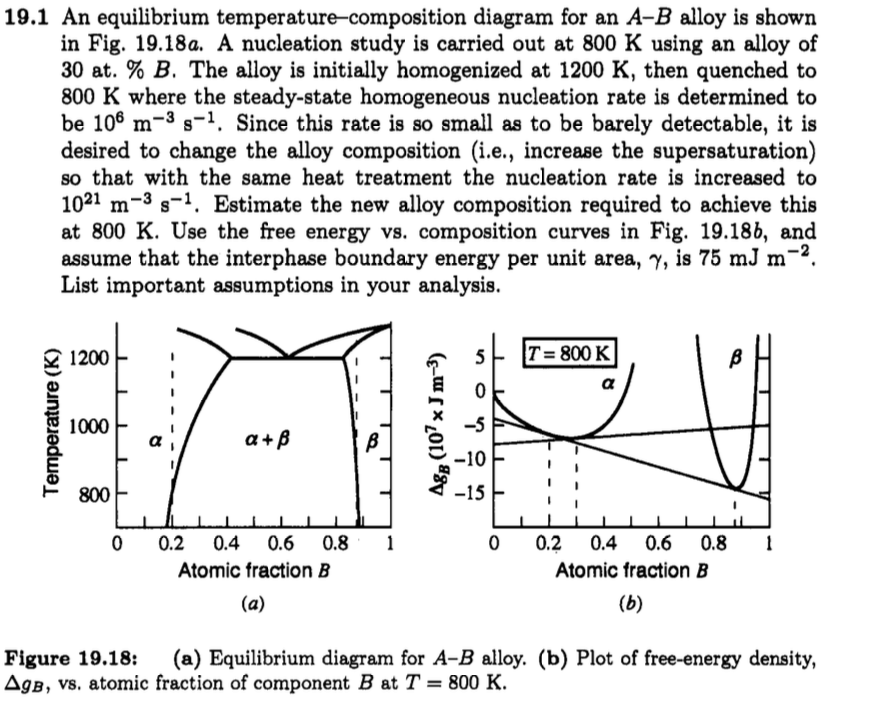
Nucleation demo: how do we get the values?

Nucleation demo: supersaturation limit

Summary
- Nucleation is a type of discontinuous phase transformation that is triggered by the difference in free energy at supercooling / supersaturation
- At unsteady-state conditions, nucleation free energy barrier is caused by the positive interfacial energy
- Nucleation free energy barrier is characterized by \(\Delta G_c\), giving critical nucleus size \(n_c\)
- The evolution of particle number at each size \(N_n\) can be described by a “diffusion-like” analog