MATE 664 Lecture 14
Introduction To Nucleation Theory
- Slides 👉 Open presentation🗒️
- PDF version of course note 👉 Open in pdf
- Handwritten notes 👉 Open in pdf
Learning outcomes
After this lecture, you will be able to:
- Identify the driving force when a system becomes supercooled or supersaturated
- Describe the difference between discontinuous and continuous phase transformations
- Analyze the nucleation free energy barrier \(\Delta G_c\)
- Describe the role of surface and interfacial energy in nucleation
- Describe the pseudo-steady-state kinetic model for nucleation
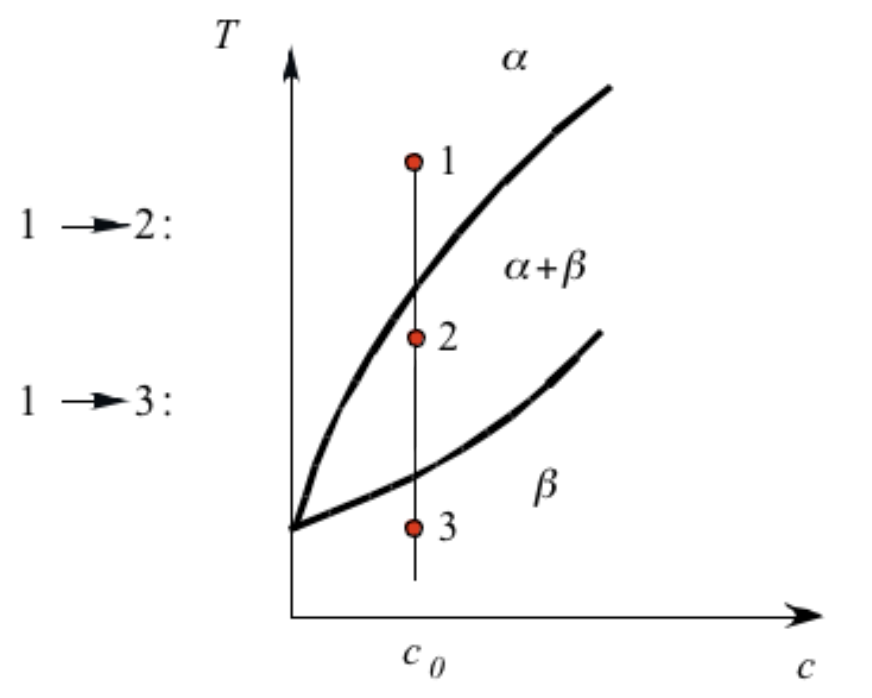
Phase-diagram in non-equilibrium region
- How to induce phase transformation from a phase diagram?
- Going low in temperature –> (super)cooling
Transformation driving force in single-component phase diagram
- Cooling from liquid to solid provides driving force
\[ \Delta G^{\text{L->S}} = \frac{\Delta H^{\text{L->S}}(T_m - T )}{T_m} < 0 \]
- Why doesn’t ice form spontaneously?
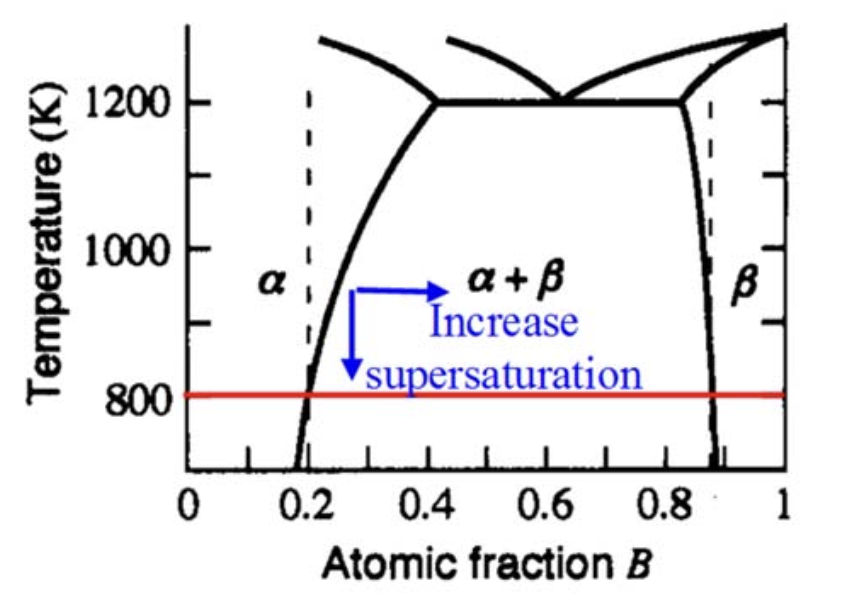
Phase transformation in binary phase diagram (\(T-X_B\))
Ways to introduce driving force:
- (Super)saturation
- (Super)cooling
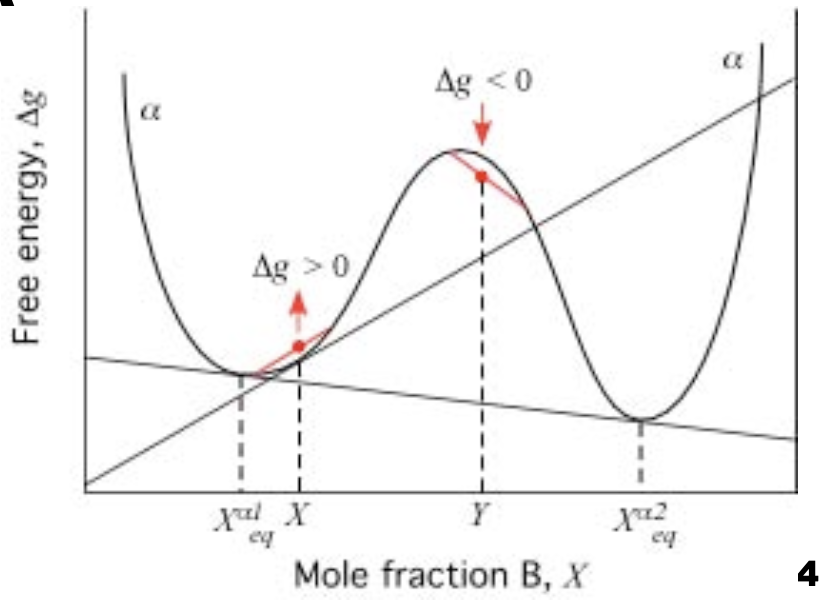
Phase transformation in binary phase diagram (\(G-X_B\))
- Metastable regions in binary phase diagram (nucleation)
- Unstable regions (spinodal decomposition)
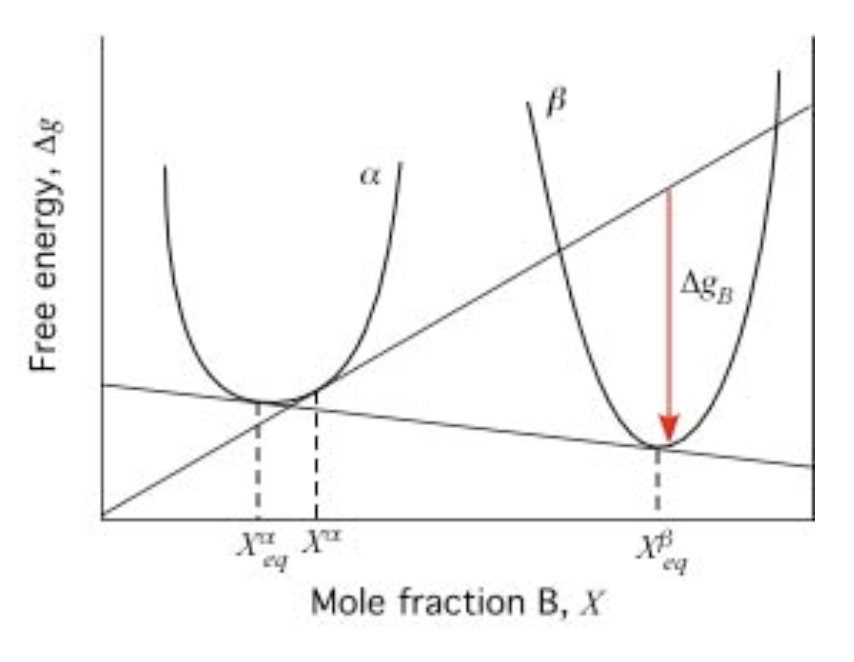
Phase transformation in binary phase diagram (\(G-X_B\))
- Metastable regions in binary phase diagram
- Tangent-to-line method: driving force \(\Delta G_B\) (molar)
Nucleation theory in a nutshell
- Crystal growth can be divided in 4 regions
- Nucleation theory deals with regions I and II (incubation & pseudo-steady-state)
Introducing interfacial energy (1)
Creating interfaces between different atoms causes energy to change!
Surface energy \(\gamma_A\) (vacuum, unit J/m\(^2\) or N/m)
\[ \gamma_A = \frac{1}{2 a_0} w_{AA} (Z_s - Z_b) \]
Interatomic energy in bulk
Creation of surface
Introducing interfacial energy (2)
Interfacial energy \(\gamma_{AB}\) can be calculated using \(\gamma_A\) \(\gamma_B\) and \(\Delta W_{AB}\)
If adhesion between A and B are not strong, interface unlikely to form!
\[ \gamma_{AB} = \gamma_A + \gamma_B - \Delta W_{AB} \]
Nucleation theory: overall nucleation free energy
- Nucleation free energy has bulk and interface parts
- Shape factor \(\eta = (36 \pi)^{1/3} \Omega^{2/3}\)
Different scaling between bulk & interfacial F.E.
- \(\Delta G^{\text{bulk}} \propto -n^{1.0}\)
- \(\Delta G^{\text{interfacial}} \propto +n^{2/3}\)
- Maximum \(\Delta G_c\) at \(n_c\).
- \(\partial G_c/\partial n |_{n=n_c} = 0\)
The critical nucleus size \(n_c\)
Classical homogeneous nucleation gives
- Critical nucleus size
\[ n_c = -\frac{8}{27} \left[\frac{\eta \gamma_{\alpha \beta}}{\mu_\beta - \mu_\alpha} \right]^3 \]
- Nucleation free energy barrier
\[ \Delta G_c = \frac{4}{27} \frac{(\eta \gamma)^3}{(\mu_\beta - \mu_\alpha)^2} \]
Pseudo-steady-state nucleation theory
- Consider the “quasi-diffusion” in \(n\)-landscape!
Q.S.S. governing equations
- “Quasi flux” of \(n \rightarrow n+1\): \(J_n\)
- Can also model \(\partial N_n/\partial t\) from assignment 2
- At equilibrium, detailed balance follows:
\[ J_n(t) = \beta_n N_n(t) - \alpha_{n+1} N_{n+1}(t) = 0 \]
- The nucleation rate at quasi-pseudo-state can be computed using a known \(N_n\) distribution
Q.S.S. assumptions
- In a constrained equilibrium system, \(N_n\) follows the Boltzmann distribution
\[ \frac{N^{\text{ceq}}_n}{N_t} \approx \exp \left( -\frac{\Delta G_n}{k_B T} \right) \]
- Result:
\[ J_n(t) = - \beta_n \left[ \frac{\partial N_n}{\partial n} + \frac{N_n}{k_B T} \frac{\partial \Delta G_n}{\partial n} \right] \]
- Analog: diffusion in external potential
\[ J = -L_{11} \nabla (\mu_1 + \phi) = -D_1 \left( \frac{\partial c}{\partial x} + \frac{c}{k_B T}\frac{\partial \phi}{\partial x} \right) \]
Q.S.S. nucleation rate: final results
- Assuming the nucleation rate is determined by \(J \approx J_{n_c}\)
- \(Z\) is the Zeldovich factor (~0.1)
Implication of Q.S.S. nucleation rate
- Zeldovich factor is around 0.1
- Particles can shrink when they are not reaching \(n_c\)!
- Rule of thumb: \(\Delta G_c \leq 76 k_B T\), otherwise no detectable nucleation
- At \(T=298\) K, \(\Delta G_c \leq 1.95\) eV
Summary
- Nucleation is a type of discontinuous phase transformation that is triggered by the difference in free energy at supercooling / supersaturation
- At unsteady-state conditions, nucleation free energy barrier is caused by the positive interfacial energy
- Nucleation free energy barrier is characterized by \(\Delta G_c\), giving critical nucleus size \(n_c\)
- The evolution of particle number at each size \(N_n\) can be described by a “diffusion-like” analog
What to learn next
Is homogeneous nucleation the whole picture? Maybe not. Consider the following examples
Sugar crystal formation
- Heterogeneous nucleation

Snow formation
- Diffusion-controlled growth
