MATE 664 Lecture 18
Growth Phenomena: Stability and Aggregation In Colloids
- Slides 👉 Open presentation🗒️
- PDF version of course note 👉 Open in pdf
- Handwritten notes 👉 Open in pdf
Learning outcomes
After this lecture, you will be able to:
- Describe the thermodynamic and kinetic stability of colloids
- Recall the balance between thermodynamic and kinetic factors
- Identify aggregation phenomena in colloids, including DLCA and RLCA
- Analyze the dimensionality of DLCA and RLCA aggregates
- Describe the stability and kinetic barrier of charged colloids
Growth mechanism in colloids
Colloids serve as a good platform for studying the kinetic phenomena we learned in this course:
- Stability: colloids may be thermodynamically unstable but kinetically stable (why?)
- Growth: can we use our kinetic theory of coarsening to predict size distribution of colloidal particles?
- Aggregation: similar to the coalescence in solid materials, how do colloidal particles merge with each other?
Colloidal example 1: Faraday’s gold solution
In 1850s, Faraday found that chemical treatment of thin gold films produced ruby colored liquid that scattered light. The liquid, kept in the basement of the Faraday Museum in London, is still stable up to today!

Colloidal example 2: milk curdling / casein coagulation
All the cheese we eat today comes from the same procedure when the colloidal suspension in milk aggregates into a dense cluster due to lactic acid produced by bacteria.

The stability statement of colloids
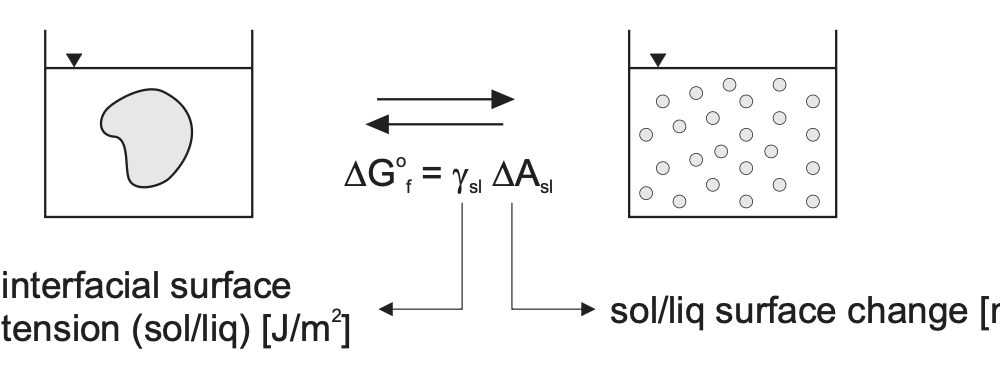
Making a bulk material (solid-state or polymer) into small particles increases the total surface area. The thermodynamic stability is determined solely by \(\gamma_{SL}\)
- \(\gamma_{SL} > 0\): unstable colloid state (lyophobic). E.g. Faraday’s solution
- \(\gamma_{SL} < 0\): colloid state (lyophilic). E.g. micelles
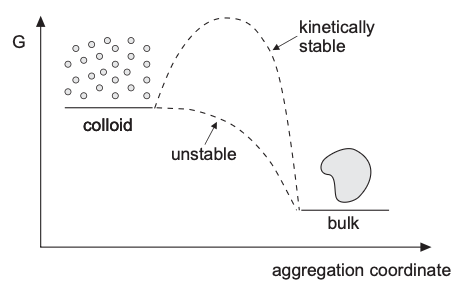
Topic 1: how can unstable colloids become kinetically stable?
Lyophobic (unstable) colloids can be made kinetically stable by building an energy barrier sufficiently large with respect to \(k_BT\).
Two stabilization mechanisms are possible:
- electrostatic: the particles are electrically charged (DLVO theory)
- steric: the particles are coated with some material (e.g. polymer) which prevents their close approach.
Topic 2: how do unstable colloids aggregate?
If the repulsive interactions between the particles are not strong enough to prevent coalescence, we will end up with the aggregation phenomenon, very similar to the coarsening:
Diffusion limited colloidal aggregation (DLCA): particles will interact with each other instantly, but flux will be limited by the diffusion
Reaction limited colloidal aggregation (RLCA): particles movement in solution is much faster, and limited by probability of sticking to each other
We will discuss about DLCA vs RLCA first in this lecture.
Prelude: mean-field treatment of colloid aggregation
Before solving the aggregation of larger particles, let’s first consider how fast individual particles stick to each other in the diffusion-limited regime.
Mean-field treatment, assume steady radial flux:
- far away: bulk number concentration \(N_0\)
- at contact: particles are absorbed \(N < N_0\)
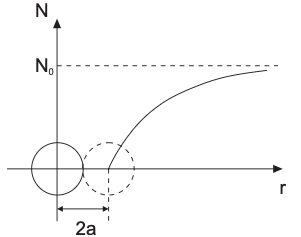
Mean-field two-particle picture: flux equations
Smoluchowski gave an expression for the two-particle aggregation flux \(F\), with mutual diffusivity \(D_{11}\) between size-1 particles.
\[\begin{align} F &= 4 \pi r^2 D_{11}\frac{dN}{dr} \\ D_{11} &= 2D \end{align}\]DLCA rate constant for primary particles
At contact, if all incoming particles are absolbed, the number density at contact distance \(R_{11}\) follows \(N(R_{11})=0\), giving the result
\[\begin{align} F &= 4 \pi D_{11} R_{11} N_0 \\ \beta_{11} &= 4 \pi D_{11} R_{11} \end{align}\]For equal spheres with radius \(a\), we have \(R_{11} = 2 a\), the coefficient \(\beta_{11}\) becomes:
\[\begin{align} \beta_{11} &= 16 \pi D a \end{align}\]Two-particle aggregation rate equation (1)
Since the particles are moving in liquid, Stokes-Einstein equation can be used to express single-particle \(D\):
\[\begin{align} D &= \frac{kT}{6\pi\eta a} \end{align}\]We get the coefficient \(\beta_{11}\) to be independent of size
\[\begin{align} \beta_{11}^{\mathrm{DLCA}} &= \frac{8kT}{3\eta} \end{align}\]Two-particle aggregation rate equation (2)
The number concentration flux \(F\) (unit s\(^{-1}\)) tells how frequent a foreign particles enters the perimeter of the primary one. The total number of aggregated particles per second, \(R_{\text{agg}}^{0}\) follows:
\[\begin{align} R_{\text{agg}}^{0} &= - \frac{1}{2}N_0 F \\ &= -\frac{1}{2} 4 \pi D R_{11} N_0^2 \\ &= -\frac{1}{2}\beta_{11} N_0^2 \end{align}\]- The factor \(1/2\) counts for de-duplication
- \(R_{\text{agg}}^{0} \propto \beta_{11} N_0^2\): looks like a second-order kinetic rate reaction
Key DLCA message
- \(\beta_{11}^{\mathrm{DLCA}}\) is independent of particle size
- aggregation is controlled by thermal transport and viscosity
- the faster the diffusion, the faster the coagulation
- this gives a natural upper bound for aggregation kinetics
Size dependence for unequal particles
If particle \(i\) and \(j\) have unequal radii \(R_i\) and \(R_j\), \(\beta_{ij}\) becomes:
\[\begin{align} \beta_{ij} &= 4\pi (D_i + D_j)(R_i + R_j) \\ &= \frac{2k_B T}{3\eta} \left(R_i + R_j\right) \left(\frac{1}{R_i} + \frac{1}{R_j}\right) \end{align}\]- “aggregation coefficient” \(\beta_{ij}\) depends on the particle size ratio!
- \(\beta_{ij}\) rank: large-small > large-large ~ small-small (why?)
DLCA rate constant: size mismatch
Assume the primary size \(a_i\) is constant, reducing \(a_j\) will monotonically crease the coefficient (favour large-small)

- this indicates the DLCA cluster shape may be very sparse (see later)
From pair collisions to a population balance
Let \(N_k\) be the number concentration of clusters of mass \(k\), how do each cluster change over time (similar to the coarsening case)?
- Smoluchowski population balance equation
- first term: birth of clusters of size \(k\)
- second term: loss of clusters of size \(k\)
Constant-kernel DLCA result (\(\beta_{ij}=\beta_{11}\))
If \(\beta_{ij} = \beta_{11}\) is constant, then the total number concentration (\(N = \sum N_k\)) obeys
\[\begin{align} \frac{dN}{dt} &= -\frac{1}{2}\beta_{11}N^2 \end{align}\]that looks like our two-particle solution, with the total number of particles decrease over time:
\[\begin{align} \frac{1}{N} = \frac{1}{N_0} + \frac{\beta_{11}}{2}t \end{align}\]Characteristic coagulation time scale
Since the population balance follows a second-order reaction \(N/N_0 \propto t^{-1}\), the characteristic time for rapid aggregation \(\tau_{\mathrm{RC}}\) follows (usually in milliseconds):
\[\begin{align} \tau_{\mathrm{RC}} &= \frac{2}{\beta_{11}N_0} \\ &= \frac{3\eta}{4k_B T\,N_0} \end{align}\]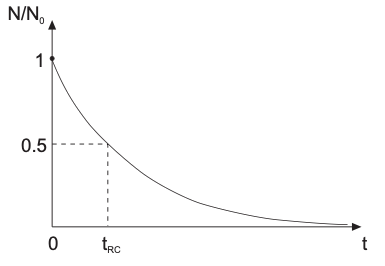
What’s missing in the current picture?
- \(\beta_{ij}\) treatment, is it really constant?
- morphology matters: aggregated clusters contain voids
- the shape of a cluster may not be a sphere!
We can solve these problem by introducing the fractal dimensionality \(d_f\), so
\[\begin{align} R_i \sim i^{1/d_f} \end{align}\]Ideal case for sphere: \(d_f = 3\). We will see the \(d_f\) distinguishes the DLCA and RLCA regimes.
DLCA radius law and kernel with fractal clusters
Rationale: we want to study the population distribution with mass \(i\), \(j\), and use \(d_f\) to link them to the \(\beta_{ij}\)
\[\begin{align} \beta_{ij}^{\mathrm{DLCA}} = \frac{2k_B T}{3\eta} \left(i^{1/d_f}+j^{1/d_f}\right) \left(i^{-1/d_f}+j^{-1/d_f}\right) \end{align}\]- the radius-mass relation is the bridge from structure to kinetics
- this is the key extension beyond the constant-\(\beta\) model
DLCA scaling law with fractal dimensionality
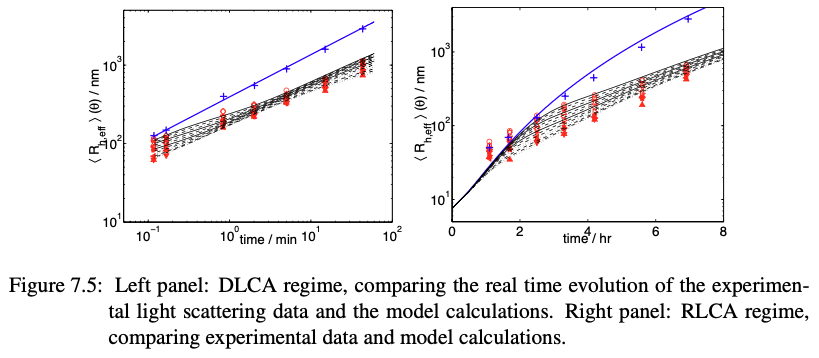
Remember we’re interested in the scaling between size \(R\) and its power law scaling to time \(t\) (like in coarsening), a useful scaling follows:
\[\begin{align} \langle R \rangle \sim t^{1/d_f} \end{align}\]- open clusters grow with a power law in time
- measuring \(\langle R_h \rangle\) vs. \(t\) gives access to \(d_f\)
- typical DLCA clusters are loose
Enter RLCA: a kinetic barrier appears
- Particles still diffuse toward each other
- A repulsive interaction creates a barrier in the total potential
- Collision becomes probabilistic (different rate law)
General picture of kinetic barrier in diffusion potential field
For a external potential \(V_{T}\) on the particle, the diffusion potential contains both chemical and external potentials:
\[\begin{align} F = 4\pi r^2 D_{11} \left( \frac{dN}{dr} + \frac{N}{k_B T}\frac{dV_T}{dr} \right) \end{align}\]- \(DLCA\) is simply \(V_T\) has no barrier
- \(RLCA\) comes into play when \(V_T\) as a maximum
Fuchs stability ratio
The reduction of aggregation rate between DLCA and RLCA is measured by
\[\begin{align} W &= \frac{\beta_{11}^{\mathrm{DLCA}}}{\beta_{11}} \\ &= 2a\int_{2a}^{\infty} \exp\left(\frac{V_T}{kT}\right)\frac{dr}{r^2} \end{align}\]which is just
\[\begin{align} \beta_{11}^{\mathrm{RLCA}} = \frac{\beta_{11}^{\mathrm{DLCA}}}{W} \end{align}\]Barrier sensitivity in RLCA
A common approximation for the Fuchs stability ratio \(W\) is:
\[\begin{align} W \approx \frac{1}{2\kappa a} \exp\left(\frac{V_{T,\max}}{k_B T}\right) \end{align}\]- a modest barrier in \(V_T\) is enough to alter the kinetics!
- how do we change the barrier? DLVO theory (next lecture)
Fractal dimensionality in RLCA
A useful form for RLCA aggregation constant is
\[\begin{align} \beta_{ij} = \frac{\beta_{ij}^{\mathrm{DLCA}}}{W} (i j)^{\lambda} \end{align}\]with
\[\begin{align} \lambda = \frac{d_f - 1}{d_f} \end{align}\]For RLCA when \(d_f \approx 2\), \(\lambda \approx 0.5\)
DLCA vs RLCA: rate comparison
Physical consequence of the RLCA kernel
DLCA
- \(W \approx 1\)
- \(\lambda = 0\)
- equal-size aggregation rate is nearly mass independent
- big-small collisions are favored
RLCA
- \(W \gg 1\)
- \(\lambda \approx 0.5\)
- equal-size aggregation rate increases with mass
- big-big aggregation becomes more important
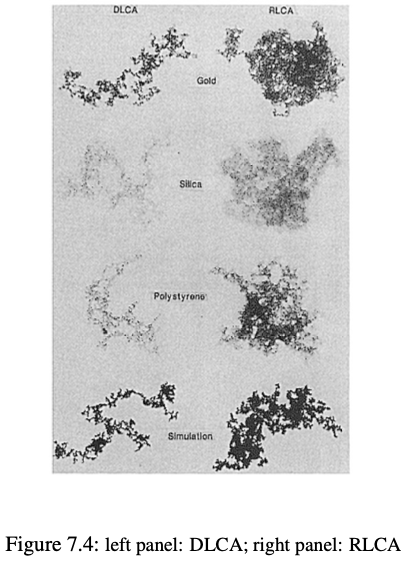
DLCA vs RLCA: structural comparison
A rule of thumb for the dimensionality \(d_f\) in polymer colloids
- DLCA: \(d_f \approx 1.6 - 1.9\)
- RLCA: \(d_f \approx 2.0 - 2.2\)
DLCA vs RLCA: time evolution
Summary
- DLCA and RLCA describe two limiting kinetic regimes of colloid aggregation
- Population-balance and two-particle models connect diffusion, sticking probability, and aggregation rate
- A kinetic barrier can strongly slow aggregation and sets up the need for DLVO theory
Comments on the time scale
\[ \tau_{\mathrm{RC}} = \frac{3\eta}{4k_B T N_0} \approx 2 \times{} 10^{11} \frac{1}{N_0}\quad \text([s]) \]