CO2 system: x_1 = 1.406e-04, y_2= 0.200
SO2 system: x_1 = 1.592e-02, y_2= 0.159CHE 318 Lecture 23
Mass Transfer In Two-Phase Column: Operating Line
- Slides 👉 Open presentation🗒️
- PDF version of course note 👉 Open in pdf
- Handwritten notes 👉 Open in pdf
Learning outcomes
After this lecture, you will be able to:
- Recall the overall mass-balance framework for two-phase absorption systems.
- Identify the operating line on an equilibrium diagram.
- Apply equations to calculate outlet composition and minimum flow rate for an absorption tower.
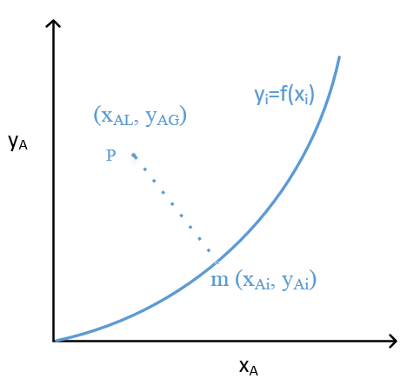
Recall: reading an equilibrium diagram
Key features:
- x-axis & y-axis meaning?
- Points on the eq. curve?
- Points above and below eq. curve?
- How to get interfacial composition?
- Slope of line to interfacial points?
Local overall mass transfer coefficients
- Knowing \(k_x\) and \(k_y\) (film coefficients) allows us to calculate the interfacial concentration \((x_{Ai}, y_{Ai})\)
- However, in many industrial applications exact \(k_x\) and \(k_y\) are hard to find
- Use of overall mass transfer coefficients
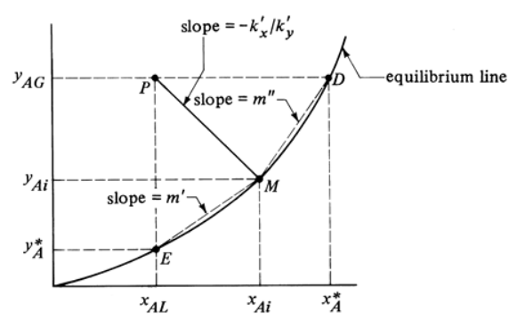
Relation between overall and exact mass transfer coefficients
- Exact coefficient \(k_x\), \(k_y\): driving force \(y_{AG} - y_{Ai}\)
\[ N_A = k_y(y_{AG} - y_{Ai}) \]
- Overall coefficient \(K_x\), \(K_y\): driving force \(y_{AG}-y_A^*\)
\[ N_A = K_y (y_{AG} - y_A^*) = K_x (x_A^* - x_{AL}) \]
\(K_x\) and \(K_y\) on a diagram
- What are the driving forces associated with \(K_x\) and \(K_y\)?

\((x, y)\) relation in a reactor
- Consider an absorption tower with gas inlet at \(y_{A1}\) and outlet at \(y_{A2}\)
- The gas solute is continuously absorbed by the flowing water phase
- Which is larger, \(y_{A1}\) or \(y_{A2}\)?
Absorption operating line in equilibrium diagram
- Mass balance tells us \(y_{A1} > y_{A2}\)
- A series of \((x, y)\) points during the operation forms the operating line
What questions do we want to study?
Given information about the absorption tower, can we answer?
- In- and out-let molar fractions ✅
- Required liquid / gas flow rate ✅
- Concentration profile → needs to know \(N_A\)
- Height of tower needed → needs to know \(N_A\)
Mass balance in whole control volume
Mass balance for 2 phases:
\[\begin{align} \text{In}_{\text{liq}} + \text{In}_{\text{gas}} &= \text{Out}_{\text{liq}} + \text{Out}_{\text{gas}} \\ L_2 x_2 + V_1 y_1 &= L_1 x_1 + V_2 y_2 \end{align}\]- \(L_1 = L' + L_{x1}\) (\(L'\): flow rate inert liquid)
- \(V_1 = V' + V_{y1}\) (\(V'\): flow rate inert gas)
Explanation for flow rates
Other flow rates: \(Q\) (m\(^3\)/s); \(W\) (kg/s); \(v\) (m/s)
- \(L\): molar flow rate (kg mol/s) for liquid phase
- \(L'\): flow rate for inert liquid
- \(L_{x1}\): flow rate for A at molar fraction \(x_1\)
- \(V\): molar flow rate (kg mol/s) for gas phase
- \(V'\): flow rate for inert gas
- \(V_{y1}\): flow rate for A at molar fraction \(y_1\)
Mass balance for operating line
The two ends of the operating line \((x_1, y_1)\) and \((x_2, y_2)\) follow:
\[\begin{align} L'\left(\frac{x_2}{1 - x_2}\right) + V'\left(\frac{y_1}{1 - y_1}\right) = L'\left(\frac{x_1}{1 - x_1}\right) + V'\left(\frac{y_2}{1 - y_2}\right) \end{align}\]Meaning of the operating line
When \(1 - x_1 \approx 1\) and \(1 - y_1 \approx 1\), we can rewrite the mass balance equation for any \((x, y)\) along the operating line
Absorption tower design requirements
In absorption tower, we usually know the following quantities:
- Gas inlet fraction \(y_1\) and flow rate \(V_1\)
- \(V'\) can be calculated
- Liquid inlet fraction \(x_2\) (usually \(x_2=0\))
- The equilibrium curve \(y=f_{\text{eq}}(x)\)
One of the following design goals may be asked:
- Know the flow rate \(L'\) → Determine \(y_2\)
- \(y_2\) needs to be at certain value → Determine \(L'\)
Minimum operating line for absorption tower
For question 2, we know the requirement for \(y_2\), combine with the equilibrium chart, there is a minimum liquid flow rate \(L'_{\text{min}}\).
Flow rate in absorption tower
From the dilute regime operating line, the slope is determined by \(\left(\dfrac{L'}{V'}\right)\). Although there is a minimal \(\left(\dfrac{L'}{V'}\right)\) requirement, practical operating line has slope that follows
\[ \left(\dfrac{L'}{V'}\right) \approx 1.5 \times \text{[Slope of Eq. Curve]} \]
- If \(L'/V'\) is too high, the column usually needs a larger diameter to compensate the pressure drop.
- If \(L'/V'\) is too low, a taller absorption tower is needed for sufficient contact area.
Example 1: determine the outlet composition
A mixture of gas A in air kept at total pressure of 1 atm flows through an absorption tower with flowing water at 293 K. The inlet gas flow rate is \(V_1 = 100\) kg mol/h, and inlet \(y_1 = 0.20\). The liquid inlet flow rate is \(L' = 300\) kg mol/h and inlet contains no dissolved gas (\(x_2=0\)). At the outlets the gas-liquid phases reach equilibrium following the Henry’s law:
\[ y_2 = m x_1 \]
Calculate the outlet mole fraction \(y_2\) and \(x_1\) for the following cases:
- A is CO\(_2\), \(m = 1.42\times 10^3\)
- A is SO\(_2\), \(m \approx 10\)
Example 2: solution process
- Write the mass balance equation
Obtain \(L'\) and \(V'\)
\(x_1\) and \(y_2\) relation from Henry’s law
Example 2: results
\(y_1\) and \(y_2\) are supposedly large, so \(1 - y_1 \approx 1\) is not correct!
Example 2: quanlitative analysis
- Slope of operating line \(L'/V'\)
- Slope of equilibrium curve \(m\)
- CO\(_2\): \(m \gg L'/V'\), can be treated as if gas phase concentration is fixed!
- SO\(_2\): measurable decrease of SO\(_2\) fraction in outlet gas
Summary
- Reading equilibrium chart and operating line
- Mass balance equation for 2 phases
- Solving the flow rate – concentration relation