CHE 318 Lecture 32
Cooling Tower Design (III)
- Slides 👉 Open presentation🗒️
- PDF version of course note 👉 Open in pdf
- Handwritten notes 👉 Open in pdf
Learning outcomes
After this lecture, you will be able to:
- Recall the form of the cooling-tower height equation from coupled heat- and mass-transfer balances.
- Describe interfacial enthalpy driving forces using the enthalpy-temperature chart.
- Apply the design procedure to estimate tower height for a representative cooling-tower problem.
- Analyze how the model can be extended to predict the bulk-gas temperature profile.
Cheatsheet for cooling tower
Recap: interfacial energy transfer
We wanted to solve the differential form
\[\begin{align} L c_L dT_L &= h_L a (T_L - T_{Li}) dz \end{align}\]- L.H.S.: sensible heat in liquid \(L c_L dT_L = h_L a (T_L - T_{Li}) dz\)
- R.H.S.: sensible + latent heat in gas
Recap: solving the energy transfer in gas-phase
For the latent heat \(q_{G, \lambda}\), it is solved by
\[\begin{align} q_{G, \lambda} &= M_A N_A \lambda_0 \\ &= M_A k_y a dz (y_i - y) \lambda_0 \\ &\approx M_A k_G a P dz \frac{M_B}{M_A}(H_i - H) \lambda_0 \\ &= M_B \lambda_0 k_G a P dz (H_i - H_G) \end{align}\]- The derivation for \(q_{G, \lambda}\) uses the fact \(y \approx \frac{M_B}{M_A} H\)
- Pressure-based coefficient \(k_G a\) often used instead of \(k_y a\)
Energy transfer in liquid: final results
Adding the sensible & latent heat in liquid side gives
\[\begin{align} G dH_y &= q_{G,S} + q_{G,\lambda} \\ &= h_G a dz (T_i - T_G) + \lambda_0 a N_A M_A \\ &= h_G a dz (T_i - T_G) + M_B \lambda_0 k_G a P dz (H_i - H_G) \end{align}\]As can be expected, both temperature and humidity driving forces should exhist.
Energy transfer in liquid: adiabatic process
Since the evaporation at interface is similar to the adiabatic process, the following relation (see Lecture 29) can be used:
\[ \frac{h_G a}{M_B k_y a} = \frac{h_G a}{M_B k_G P} \approx c_s \]
which gives heat transfer in gas phase as
\[\begin{align} G dH_y &= c_s M_B k_G a P dz (T_i - T_G) + M_B \lambda_0 k_G a P dz (H_i - H_G) \\ &= M_B k_G a P dz \left[ c_s (T_i - T_G) + \lambda_0 (H_i - H_G) \right] \\ &= M_B k_G a P dz (H_{yi} - H_y) \end{align}\]Heat transfer at interfaces: implications
What are the implications for the follong equation?
\[\begin{align} G dH_y &= M_B k_G a P dz (H_{yi} - H_y) \end{align}\]- The enthalpy in the gas phase \(H_y\) has an associated “transfer coefficient” \(M_B k_G a P\)!
- That justifies our choice of Enthalpy - Temperature chart.
- Enthalpy driving force in gas phase
- Temperature driving force in liquid phase
Interfacial flux equation for cooling tower
Combining the L.H.S with R.H.S we get
\[\begin{align} M_B k_G a P dz (H_{yi} - H_y) &= h_L a (T_L - T_{Li}) dz \\ \frac{H_{yi} - H_y}{T_{Li} - T_{L}} &= -\frac{h_L a }{M_B k_G a P} \end{align}\]- The slope to find interfacial \((H_{yi}, T_{Li})\) is \(-\frac{h_L a }{M_B k_G a P}\)
- No longer need to do iterative slope searching, only 1 calculation!
Link to humidity chart adiabatic saturation curves
Recall in Lecture 29, the slope of adiabatic curves in psychrometric chart is given by
\[ \frac{H_w - H}{T_w - T} = - \frac{h_G}{M_B k_y \lambda_w} \]
On the other hand the slope to find interfacial \((H_{yi}, T_{Li})\) in cooling tower is
\[ \frac{H_{yi} - H_y}{T_{Li} - T_{L}} = -\frac{h_L a }{M_B k_G a P} \]
They have very similar forms, but be careful one is purely in gas phase and the other describes the 2-phase equilibrium.
Solving the tower height
If only use the R.H.S result
\[\begin{align} G dH_y &= M_B k_G a P dz (H_{yi} - H_y) \end{align}\]we can obtain the total tower height by integration
\[\begin{align} Z &= \int_{0}^{Z} dz \\ &= \int_{H_{y1}}^{H_{y2}} \frac{G}{M_B k_G a P} \frac{dH_y}{H_{yi} - H_y} \end{align}\]The integral is carried out over \(d H_y\), not \(T_L\)!
Steps to solve the cooling tower
Similar to absorption tower, cooling tower design typically involves the following steps
- Plot the saturated air enthalpy \(H_{yi}\) vs liquid temperature \(T_L\) (usually a given chart)
- Knowing the entering air conditions \(T_{G1}\) and \(H_1\) (humidity), calculate the enthlpy \(H_{y1}\) (not saturated).
- The tower bottom \((T_{L1}, H_{y1})\) on the operating line is determined from \(T_{L1}\) requirement
- Find the minimal gas flow rate \(G_{min}\) and practical operating \(G\), determine tower top operating line point \((T_{L2}, H_{y2})\)
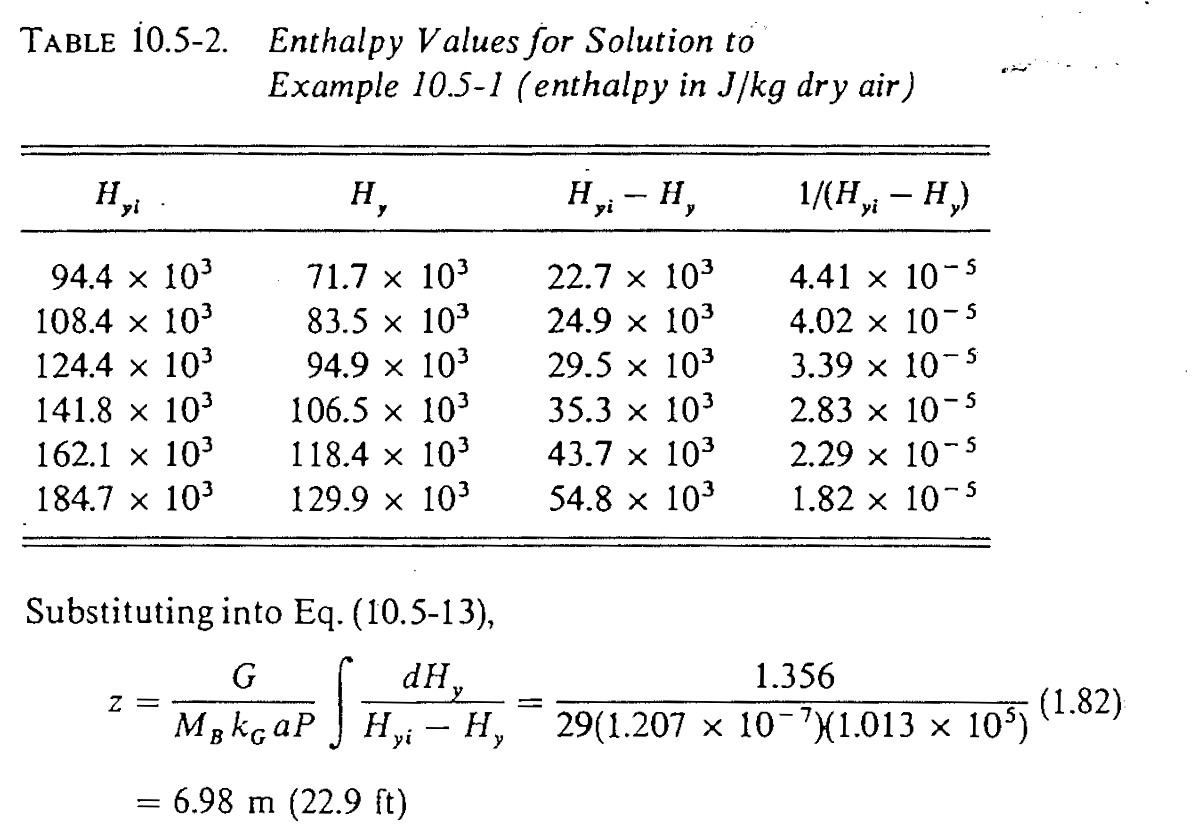
- Know the \(h_L a\) and \(k_G a P\) (or \(k_y a\)) values, pick several points along the operating line, use slope \(-\frac{h_L a }{M_B k_G a P}\) to find \(H_{yi}\) and calculate \(1 / (H_{yi} - H_y)\)
- Numerically integrate \(\int_{H_{y1}}^{H_{y2}} 1 / (H_{yi} - H_y) dH_y\) and \(\frac{G}{M_B k_G a P}\) to find \(Z\)
Cooling tower design problem example

A similar example is given in Assignment 8. Be careful about the units!
Finding the humidity of inlet air
Interfacial demo
Textbook solution (1)

Textbook solution (2)
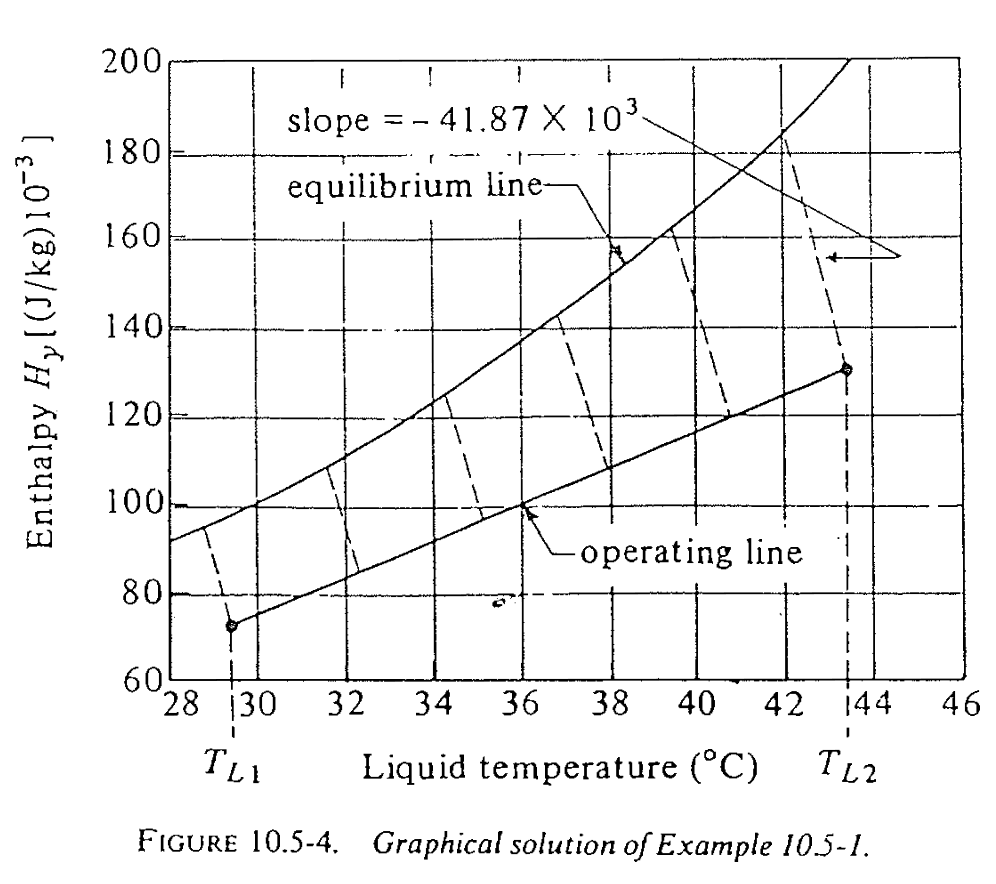
Textbook solution (3)
We get \(Z = 7.03\) m, pretty close!
Final question: what if we wanted to know the \(T_G\) as well?
Advanced discussion. May not appear in final exam.
For the change of bulk-gas temperature \(T_G\), the following energy flux equation can be given for the sensible heat in gas \(q_{G, S}\):
\[\begin{align} c_s G dT_G &= h_G a dz (T_{Gi} - T_{G}) \\ &= h_G a dz (T_{Li} - T_{G}) \end{align}\]For comparison we also have the change of bulk liquid temperature expressed as
\[\begin{align} c_L L dT_L &= M_B k_G a P dz (H_{yi} - H_{y}) \end{align}\]Our goal is to find a differential equation so that \(T_G\) can be integrated from \(T_{G1}\) (cool air intake)
Solving the ODE for \(T_G\)
Combine the energy balance equations for \(T_G\) and \(T_L\) gives
\[\begin{align} \frac{c_L L}{c_s G} \frac{dT_L}{dT_G} &= \frac{M_B k_G a P}{h_G a} \frac{H_{yi} - H_{y}}{T_{Li} - T_G} \\ d T_G &= \frac{c_L L}{G} \frac{T_{Li} - T_G}{H_{yi} - H_{y}} d T_L \end{align}\]- Again, we cancel out \(c_S = \frac{h_G a}{M_B k_G a P}\). This is an
- integrable ODE, given that at any \((T_L, H_y)\), we can calculate \(H_{yi}\) without needing \(T_G\)
Integrating \(T_G\): graphical explanation
Summary
- Cooling-tower height can be obtained by integrating the interfacial enthalpy driving force across the column.
- The enthalpy-temperature chart provides the graphical information needed to evaluate interfacial states.
- The same framework can be extended to estimate the bulk-gas temperature profile when needed.